

Project from 3D constructor - https://dubina.biz




An electric battery is a device that can receive electrical energy from an external source and then give it back. The process by which electrical energy is communicated to the battery is called charging the battery. The process by which the battery gives electrical energy to an external circuit is called discharging the battery. During discharging, electrical energy is obtained due to the chemical energy of the active substances formed during the charging process, and during discharging, the reverse chemical process occurs. During charging, the chemical composition of the battery plates changes, and chemical energy accumulates in them. When electrical energy is given off (during discharging), chemical processes occur in the opposite direction and the plates of the discharged battery acquire their original state (before charging), i.e. the same chemical composition. The battery does not destroy the materials included in the battery, reversible chemical reactions occur. This is how the battery converts chemical energy into electrical energy. The battery consists of an anode (+) and a cathode (-). A battery is characterized by its capacity, i.e. the amount of electrical energy that the battery can deliver when discharging from a fully charged state to the maximum permissible discharge. The capacity of a battery is measured in ampere-hours and depends on its design, the number of plates, their thickness, the material of the plate separators, and other factors. In operation, the capacity of a battery depends on the discharge current, the temperature of the electrolyte, the discharge mode (intermittent or continuous), the state of charge, and the wear of the battery. Thus, with an increase in the discharge current and a decrease in the temperature of the electrolyte, the capacity of the battery decreases. The capacity of the battery C depends on the surface area of the electrode S, the distance between the electrodes d, and the electrical conductivity of the electrolyte. The greater the surface area of the electrode S, the greater the capacity of the battery.
C ~ S,
The smaller the distance between the electrodes d, the greater the capacity of the battery.
C ~ 1 / d,
The greater the conductivity of the electrolyte, the greater the capacity of the battery.
C ~ ε,
General formula for battery capacity.
C = εε0 S / d,
Lithium batteries have high energy density and voltage, long service life and low self-discharge, they are commercially available and widely used. The car uses lithium-ion batteries, which have low resistance and are capable of delivering a current of several hundred amperes for several seconds. The battery consists of highly efficient, high-energy cylindrical cells with lithium-ion (Li-ion) chemistry of the NMC (nickel-manganese-cobalt) type with a graphite anode and a lithium cobalt cathode. The battery consists of 16 modules, each with a nominal voltage of 25 V. Sixteen battery modules are connected in series to form a battery with a nominal voltage of 400 V. Each battery module consists of 360 18650 batteries, which are very similar in appearance to simple finger batteries (the weight of one battery is 45 g), which are connected according to the 6s60p scheme: 6 elements in series and 60 such groups in parallel. The energy capacity of each battery module of such elements is 5.3 kW h. In total, the battery has 5760 such batteries. For ease of use, the battery has a voltage of 400 V. This is achieved by connecting the batteries in a mixed manner. Parallel connection of batteries is used to obtain a higher current at the same voltage. Series connection of batteries is connected in a battery alternately - the plus of one battery with the minus of another is used to obtain a higher voltage at a constant current. The total current in this case will be equal to the maximum permissible for each element. With a mixed connection, the elements are connected in parallel into groups, which are then connected in series. When connecting elements into a battery, it is necessary to remember that the battery will work well only if all the elements included in it have the same capacity and voltage. Mismatch of the parameters of individual elements, especially when connected in parallel, can lead to premature failure of the entire battery and a decrease in its efficiency. The battery is protected from the environment by a carbon fiber case. On the inside of the carbon fiber cover there are pads in the form of a film. The cover is fastened with screws with rubber gaskets, which are sealed additionally with silicone sealant. The battery block is divided into 16 compartments, each compartment contains a battery module. Each of the 16 battery modules has a built-in BMU unit, which is connected to the common BMS system, which controls the operation, monitors the parameters, and also provides protection for the entire battery. The battery has two terminals: positive and negative. The common output terminals (terminal) are located at the rear of the battery block. The battery is attached to the monocoque and the rear frame of the car using screws. It powers all devices and motors of the car. The battery has a small internal resistance and is capable of giving a current of several hundred amperes for several seconds, instantly creating power on the electric motors.
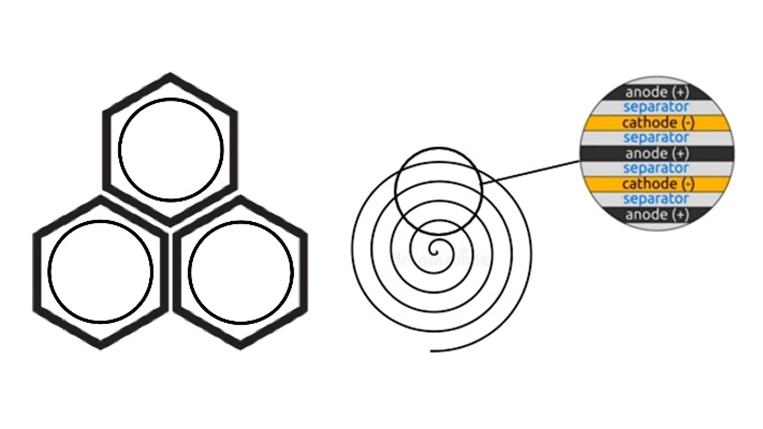
Design and arrangement of batteries in modules.
The value of the electromotive force (emf) - the voltage at its terminals when there is no load - does not depend on the size and shape of the plates, but depends only on the design of the battery. The voltage at the terminals depends on the value of the load resistance and the internal resistance of the battery. The greater the value of the internal resistance of the battery and the lower the load resistance, the lower the voltage at its terminals. Internal resistance is made up of the resistance of the electrolyte and electrodes and is measured in ohms. Capacity is the ability of the battery to give off a certain amount of electricity over a certain period of time, it is measured in ampere-hours (Ah). The value of capacity depends on the magnitude of the discharge current, the density of the electrolyte and the ambient temperature. Therefore, it can fluctuate greatly. Efficiency is the ratio of the energy given off by the battery during discharge to the energy that was supplied to it during charging, the efficiency can never be equal to one (part of the energy during charging is wasted on useless heating of the plates and electrolyte, since they have a certain resistance). The battery is characterized by the value of permissible charging and discharging currents. Exceeding these currents leads to premature failure of the battery. The battery charging rate decreases. This occurs due to the increase in electron density. The battery is charged from a charger with a constant current. The charging current does not exceed half the battery capacity. The battery is charged unevenly. At first, the battery charges quickly, then more slowly. This occurs due to the increase in charge density in the battery. During charging, the number of positively charged LIC6 molecules on the anode increases, their density becomes higher. The resistance and voltage in the battery increase. To fully charge the battery, more energy is required to form new molecules. The graph of the battery charge versus time has the form of a function y = x ^ 1 / 2.
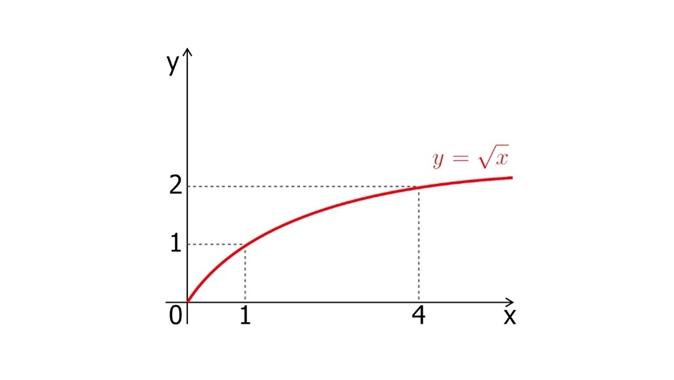
Charging speed versus time graph.
The battery cathode is made of an aluminum current collector with LiNiMnCoO2 applied to it. Aluminum is used as a cathode current collector, since aluminum has high electrical conductivity, low weight and low cost compared to other materials. The cathode suspension consists of LiNiMnCoO2, a binder, a conductive additive and a solvent. The battery anode is made of graphite foil 100 μm thick. The separator is located between the cathode and the anode. A thin insulating film with high ion permeability and mechanical strength is used as a separator. The separator has a pore diameter of 0.05 μm and a thickness of 10 μm. Non-woven polypropylene is used as a separator. The battery capacity is increased by increasing the surface area of the cathode and anode. To increase the battery capacity with an unchanged plate area, their area is increased by making grooves on their surface, as well as filling the lattice plates. The battery therefore has a greater capacity than with smooth plates. The increase in the surface area of the anode made of graphite foil is carried out by a thermochemical reaction. The increase in the surface area of the cathode made of aluminum sheet is carried out in a similar way.
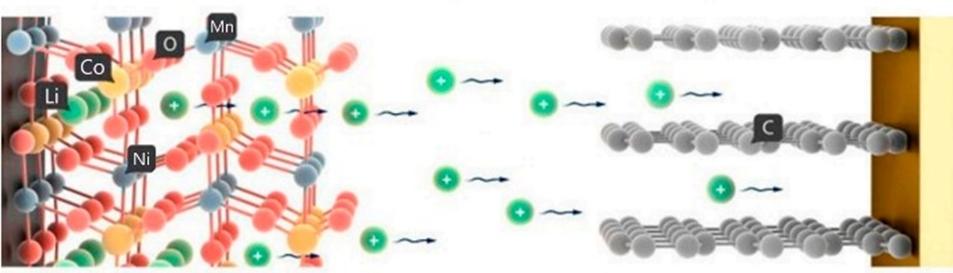
Schematic diagram of chemical processes in a battery.
Please do not open any links and do not make calls (including WhatsApp) to any numbers from messages sent by accounts such as Grabcad Verification, etc. - these are phishing ones. Please do not make any payments. Our security team is currently working on a solution.